Projects
|
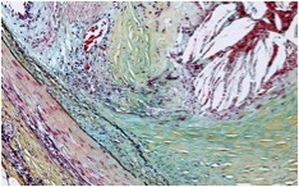
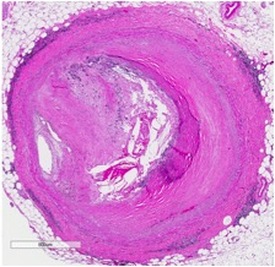
Driven by the interplay between accumulation of excess cholesterol in the arterial wall and the immune system, atherosclerosis is a disease of maladaptive inflammation. The atherosclerotic plaque grows when the rate of macrophage accumulation (via recruitment and proliferation) exceeds that of removal (e.g., via cell death and egress). The removal of excess cholesterol is intricately linked to the inflammatory status of lesions, which in turn underlies the susceptibility to plaque rupture- the ultimate clinical complication of atherosclerosis.
|
In addition to atherosclerosis, our lab investigates the role of inflammation in other metabolic diseases, including obesity. We recently uncovered a role for the RIP kinase protein RIPK1 in promoting inflammation in obesity and coronary disease in humans, and could be therapeutically targeted to reduce both of these diseases in mice.
We are now extending our work to investigate the common mechanisms of inflammation in other tissues, including the brain and liver, and will investigate how aging and other risk factors for chronic disease may alter inflammatory pathways.
We are now extending our work to investigate the common mechanisms of inflammation in other tissues, including the brain and liver, and will investigate how aging and other risk factors for chronic disease may alter inflammatory pathways.
Themes |
Theme #1: Elucidation and therapeutic targeting of pro-inflammatory microRNAs in macrophages
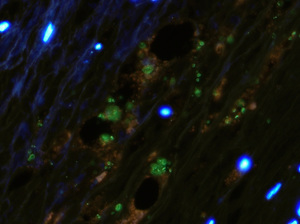
Research objectives: Over the past decade, microRNAs (miRNAs) have emerged as key modulators and fine-tuners of multiple signaling pathways involved in atherosclerosis. miRNAs are defined as highly conserved small RNA sequences of 20 to 23 nucleotides that contain complementary sequences for specific target messenger RNAs (mRNAs). Via binding to the 3’-untranslated regions (UTR), miRNAs post-transcriptionally regulate gene expression by degradation and/or translational inhibition of their bound targets. Notably, one miRNA can simultaneously repress multiple target genes through seed-based targeting. In addition, one functional gene network can be regulated by a group of miRNAs, providing a mechanism to coordinate complex gene expression programs and thereby modulate many aspects of cellular homeostasis and physiology.
Given the intersection of cholesterol and inflammation in the pathogenesis of atherosclerosis, we are investigating how inflammatory and anti-inflammatory miRNAs control atherosclerosis development by controlling myeloid-derived cells that perpetuate inflammation in the plaque. Theme #2: Investigating novel drivers of hematopoiesis in atherosclerosis
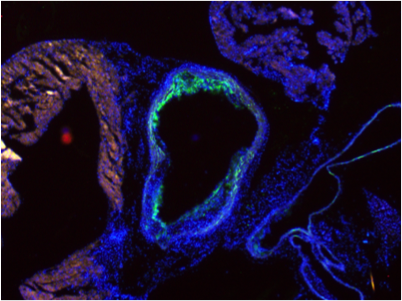
Research objectives: Although plasma LDL-cholesterol is a direct cause of atherosclerotic disease, LDL-cholesterol need not be elevated in order for atherosclerotic lesions to develop and become clinically dangerous. Indeed, there is ‘residual inflammatory risk’ present in individuals with normal lipid levels but high inflammatory indices. Circulating lipid levels are linked to inflammatory responses, and plasma lipoproteins change in response to the inflammatory needs of the host. Under steady-state conditions hematopoietic stem cells (HSCs) are quiescent, a state highly influenced by the various cell types comprising the BM microenvironment, including stromal cells and sinusoidal endothelial cells. Peripheral cues including adrenergic signaling or immune modulators signal to the microenvironment to allow for HSCs to differentiate to the various immune cells found in the circulation. However, chronic inflammatory diseases hematopoiesis can shift to peripheral sites, such as the spleen- a process known as extramedullary hematopoiesis. Additionally, during infection, hematopoiesis in sites like the spleen and liver becomes activated. While it has been demonstrated that the splenic microenvironment is unique to that of the bone marrow, there is very little known about the splenic niche and little understanding of how unique niche factors contribute to the HSC pool in the spleen. We uncovered an unexpected role for the protein MLKL as a central node in the regulation of macrophage commitment from circulating progenitors and cholesterol metabolism- all processes that drive atherosclerosis. We are now investigating these novel roles for MLKL in regulating non-necroptotic pathways during atherosclerosis.
Theme #3: Modifiable and non-modifiable risk factors of atherosclerosisResearch objectives Age is more than just a number. Age is the cumulative biological state of each cell, tissue and organ. It is a reflection of our experiences and exposures over time, the changes to our environment at the micro and macro level, and the genetic hardwiring of each cell. While not itself a disease, aging is a risk factor for many, including cardiovascular disease (CVD). Age can be chronological, defined by the unit of time since an individual’s birth, or biological, defined by the characteristics of the organism, tissue or cell relative to its baseline state. Advanced age is the single biggest risk factor for coronary artery disease and resultant heart attacks, even when other risk factors are absent1. Indeed, in individuals over 65, plaque buildup in the coronary arteries can be found even in the absence of elevated cholesterol, which is a key pathological driver of atherosclerosis. These data suggest that age as a biological factor has a clear contribution to cardiovascular disease development, and the interaction between age and other biological risks factors is not the same in older versus younger individuals..
One of the most prominent biological features of aged tissues is increased inflammation and lipid accumulation. Over time, cells acquire properties known as the “senescence-associated secretory phenotype”, or SASP, that is marked by the release of pro-inflammatory factors (TNFα, IL-6 and IL1β). During the process of senescence (growth arrest), cells have a marked production of reactive oxygen species and secretion of SASP, which induces local inflammation and tissue damage. Indeed, this low-grade persistent inflammation in aging tissues is believed to underlie much of the pathology in aged-associated diseases. Much of the focus on aging and the immune system has been to better understand the impaired responses to bacterial and viral infection leaving a major gap in our understanding of the contribution of aged immune cells to chronic inflammatory diseases like those found in the heart. Pneumonia, one of the leading causes of death worldwide, is defined as inflammation of the lungs caused by either bacteria, such as Streptococcus pneumoniae, or viruses, such as SARS-CoV2-the cause of the ongoing pandemic. Recent reports have shown hospitalization for pneumonia substantially increases the short- and long-term risk of developing cardiovascular diseases (heart attack, heart failure). The mechanism underlying this link is not well understood, however it is thought that inflammation in the lungs may spread to the rest of the body, activating systemic inflammation, even after bacterial or viral clearance. While current therapies rely on anti-inflammatory medications to inhibit the pro-inflammatory phase, these also suppresses bacterial or viral clearance. Therapies that promote the resolution of inflammation are therefore sought-after. Our studies seek to identify how factors such as age and infection can accelerate inflammatory activation in the vessel wall, and test whether existing front-line therapies are effective in this primed immune environment. |
Publications |
Selected publications:Select publications
|